Current Research
Ovarian biology
Nothing ever is, but is always becoming.
Plato, Theaetetus (circa 369 BC)
The ovary is an incredibly dynamic tissue that must protect and mature the body’s most important cell, the egg (oocyte). Around the time of birth, oocytes are surrounded by a single layer of support cells and enclosed in a basement membrane. By a mechanism that is yet to be fully understood, follicles are activated to grow one at a time from birth. Therefore, every growing follicle within an ovary is at a different developmental stage. During the growth of the ovarian follicle, the follicle increases in size and the oocyte undergoes critical maturational processes. My research group is investigating the changing microenvironment of the follicle as it grows, the relationship between the oocyte and it’s companion somatic cells and the molecular mechanisms that regulate oocyte maturation in a range of mammalian species.
We use this knowledge to underpin our research on developing novel ways to improve the efficiency of artificial reproductive technologies by identifying biomarkers for oocyte and embryo quality and refining in vitro maturation (IVM) systems.
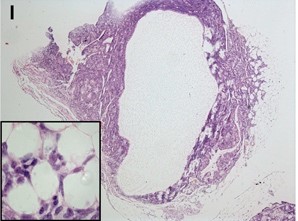
Origins of Ovarian Cancer
Most ovarian tumours exhibit an epithelial phenotype however their cell of origin remains controversial. The majority of epithelial ovarian cancers (EOC) are thought to derive from the ovarian surface epithelium (OSE). As the OSE is the primary source of somatic cells that closely associate with oocytes during ovarian follicular assembly, we have presented a new paradigm for the origin of ovarian cancer. This paradigm is based on the observation that ovarian cancers of multiple phenotypes develop following the premature loss of oocytes. This suggests that through their secretions, oocytes may suppress the multipotency of epithelial-derived ovarian cells. Using animal and in vitro models, we focus on the histological and molecular transformations that occur within the ovary from normal tissue to the development of ovarian tumours. We also have exciting new evidence that secretions from oocytes significantly suppress growth rate in a human cancer cell line. We are currently investigating the signalling pathway involved and screening for potential factors that might be responsible for this effect as a potential therapeutic tool.
Kākāpō reproduction
Kākāpō are a critically endangered parrot, endemic to New Zealand. There are now fewer than 160 individuals alive. Their low numbers are due to a combination of habitat loss, predation, disease, infertility and breeding seasons that are restricted to infrequent mass fruiting of native trees (mast years). This latter observation suggests a link between NZ native plant and kākāpō breeding. In collaboration with the kākāpō recovery team at DOC, my research group is investigating the content of phytoestrogens in native plants and fruits commonly browsed by kākāpō. We have sequenced the kākāpō oestrogen receptors and identified unique parrot-specific differences. We are using this information to refine our bioassay for measuring phytoestrogens to test whether kākāpō have a divergent sensitivity to oestrogens.

Sensitive Detection Assays using Aptamers
The accurate quantification of factors secreted into biological fluids is hampered by inadequate and insensitive techniques. In collaboration with the Aptamer team, we are generating short sequences of single-stranded DNA (aptamer) that can bind to a number of factors that are key to oocyte quality. These specific aptamers may then be used to develop sensitive and accurate detection systems for their measurement in biological fluids in a range of mammalian species.
